Read 4+ pages determine the enthalpy change for the decomposition of calcium carbonate analysis in Doc format. The Enthalpy Change of the Thermal Decomposition of Calcium Carbonate Results. Instead both calcium carbonate and calcium oxide are reacted with hydrochloric acids to. This cannot be done experimentally because it is impossible to add the add the heat required to decompose the solid and to measure the temperature change of a solid at the same time. Check also: determine and determine the enthalpy change for the decomposition of calcium carbonate 28change H for the thermal decomposition of calcium carbonate.
3 Hf 3 CO 2 -394 kJ mol-1. The Enthalpy Change of the Decomposition of Calcium Carbonate _INTRODUCTION_ RESEARCH QUESTION.

Energetics Ppt Download The formula for calculating the enthalpy change of a reaction by using the enthalpies of formation of reactants and products is.
| Topic: Determine The Enthalpy Change For The Decomposition Of Calcium Carbonate CaCO3s. Energetics Ppt Download Determine The Enthalpy Change For The Decomposition Of Calcium Carbonate |
| Content: Answer Sheet |
| File Format: Google Sheet |
| File size: 2.2mb |
| Number of Pages: 30+ pages |
| Publication Date: October 2019 |
| Open Energetics Ppt Download |
 |
13Determine the enthalpy change of the thermal decomposition of calcium carbonate by an indirect method based on Hess law.

Determine the enthalpy change for the decomposition of calcium carbonate CaCO3s CaOs CO2g given the thermochemical equations below. What is the enthalpy change of the decomposition of calcium carbonate. Calculate the enthalpy of. An Experiment to Determine the Enthalpy Change for the Decomposition of Calcium Carbonate. Record the results in a table. In this experiment Hesss law will be utilized to calculate the decomposition of Calcium carbonate.
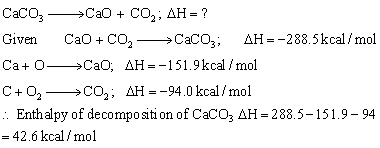
Calculate The Enthalpy For The Formation Of Calcium Chegg 5The H 0 for the decomposition of C a C O X 3 s is 1783 k J m o l at 25 C and 1000 a t m.
| Topic: This cannot be done experimentallybecause it is impossibletoadd the exactenergy required to decompose the solidand to measure the temperature changeof a solid at the same time. Calculate The Enthalpy For The Formation Of Calcium Chegg Determine The Enthalpy Change For The Decomposition Of Calcium Carbonate |
| Content: Analysis |
| File Format: DOC |
| File size: 5mb |
| Number of Pages: 40+ pages |
| Publication Date: August 2018 |
| Open Calculate The Enthalpy For The Formation Of Calcium Chegg |
 |

An Experiment To Determine The Enthalpy Change For The Deposition Of Calcium Carbonate Gcse Science Marked Teachers Of approximately 2 mol dm HCl hydrochloric acid into a polystyrene beaker using a 100cm measuring cylinder.
| Topic: CaCO3s CaOs CO2g The enthalpy change for this decomposition reaction is difficult to measure directly. An Experiment To Determine The Enthalpy Change For The Deposition Of Calcium Carbonate Gcse Science Marked Teachers Determine The Enthalpy Change For The Decomposition Of Calcium Carbonate |
| Content: Learning Guide |
| File Format: DOC |
| File size: 2.3mb |
| Number of Pages: 23+ pages |
| Publication Date: October 2021 |
| Open An Experiment To Determine The Enthalpy Change For The Deposition Of Calcium Carbonate Gcse Science Marked Teachers |
 |

13 Chapts 5 6 Test Determine The Enthalpy Change For Chegg CaOs CO2g given the thermochemical equations below.
| Topic: CaOs H2O rH 652 kJmol. 13 Chapts 5 6 Test Determine The Enthalpy Change For Chegg Determine The Enthalpy Change For The Decomposition Of Calcium Carbonate |
| Content: Answer |
| File Format: PDF |
| File size: 1.8mb |
| Number of Pages: 6+ pages |
| Publication Date: March 2018 |
| Open 13 Chapts 5 6 Test Determine The Enthalpy Change For Chegg |
 |

Investigate The Enthalpy Values For The Reaction Between Calcium Carbonate And Calcium Oxide With Hydrogen Chloride Gcse Science Marked Teachers Determining the enthalpy change for the thermal decomposition of potassium hydrogen carbonate into potassium carbonate.
| Topic: CaOs CO2g Given The Thermochemical Equations Below. Investigate The Enthalpy Values For The Reaction Between Calcium Carbonate And Calcium Oxide With Hydrogen Chloride Gcse Science Marked Teachers Determine The Enthalpy Change For The Decomposition Of Calcium Carbonate |
| Content: Explanation |
| File Format: PDF |
| File size: 725kb |
| Number of Pages: 21+ pages |
| Publication Date: September 2018 |
| Open Investigate The Enthalpy Values For The Reaction Between Calcium Carbonate And Calcium Oxide With Hydrogen Chloride Gcse Science Marked Teachers |
 |
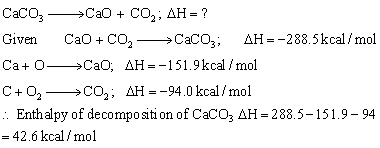
Question 9 Determine The Enthalpy Change For The Chegg CaCO s CaO s CO g HfkJmol.
| Topic: Determine the enthalpy change for the decomposition of calcium carbonate CaCO3s. Question 9 Determine The Enthalpy Change For The Chegg Determine The Enthalpy Change For The Decomposition Of Calcium Carbonate |
| Content: Answer Sheet |
| File Format: DOC |
| File size: 810kb |
| Number of Pages: 4+ pages |
| Publication Date: May 2021 |
| Open Question 9 Determine The Enthalpy Change For The Chegg |
 |

Determining The Enthalpy Change Of The Deposition Of Calcium Carbonate Gcse Science Marked Teachers CaOs H2O H.
| Topic: T1 17 T2 19 DT 02 using 257g of CaCO3 For CaO. Determining The Enthalpy Change Of The Deposition Of Calcium Carbonate Gcse Science Marked Teachers Determine The Enthalpy Change For The Decomposition Of Calcium Carbonate |
| Content: Explanation |
| File Format: Google Sheet |
| File size: 3.4mb |
| Number of Pages: 10+ pages |
| Publication Date: May 2019 |
| Open Determining The Enthalpy Change Of The Deposition Of Calcium Carbonate Gcse Science Marked Teachers |
 |

Investigate The Enthalpy Values For The Reaction Between Calcium Carbonate And Calcium Oxide With Hydrogen Chloride Gcse Science Marked Teachers 3H reactionH1-H2 This Hesss law is used to work out theenthalpy change for the thermaldecomposition of calcium carbonate.
| Topic: Question 8 a Determine the enthalpy change of reaction H in kJ when 100 mol of gaseous hydrazine decomposes to its elements. Investigate The Enthalpy Values For The Reaction Between Calcium Carbonate And Calcium Oxide With Hydrogen Chloride Gcse Science Marked Teachers Determine The Enthalpy Change For The Decomposition Of Calcium Carbonate |
| Content: Answer Sheet |
| File Format: Google Sheet |
| File size: 1.6mb |
| Number of Pages: 17+ pages |
| Publication Date: February 2018 |
| Open Investigate The Enthalpy Values For The Reaction Between Calcium Carbonate And Calcium Oxide With Hydrogen Chloride Gcse Science Marked Teachers |
 |

Calculate The Enthalpy Of Deposition Of Caco3 Into Cao And Co2the Of Formation Of Caco3 Is 2885 K Cal Mol For Cao Is 1519 K Cal Mol And For Co2is 9 Calcium carbonate is a fairly stable compound thus it must mean that more energy goes into this reaction than out to produce the desired results.
| Topic: In this experiment Hesss law will be utilized to calculate the decomposition of Calcium carbonate. Calculate The Enthalpy Of Deposition Of Caco3 Into Cao And Co2the Of Formation Of Caco3 Is 2885 K Cal Mol For Cao Is 1519 K Cal Mol And For Co2is 9 Determine The Enthalpy Change For The Decomposition Of Calcium Carbonate |
| Content: Synopsis |
| File Format: Google Sheet |
| File size: 810kb |
| Number of Pages: 13+ pages |
| Publication Date: September 2020 |
| Open Calculate The Enthalpy Of Deposition Of Caco3 Into Cao And Co2the Of Formation Of Caco3 Is 2885 K Cal Mol For Cao Is 1519 K Cal Mol And For Co2is 9 |
 |

Calculating The Enthalpy Change Of Deposition What is the enthalpy change of the decomposition of calcium carbonate.
| Topic: Determine the enthalpy change for the decomposition of calcium carbonate CaCO3s CaOs CO2g given the thermochemical equations below. Calculating The Enthalpy Change Of Deposition Determine The Enthalpy Change For The Decomposition Of Calcium Carbonate |
| Content: Analysis |
| File Format: PDF |
| File size: 1.5mb |
| Number of Pages: 11+ pages |
| Publication Date: July 2018 |
| Open Calculating The Enthalpy Change Of Deposition |
 |

Determining The Enthalpy Change Of The Deposition Of Calcium Carbonate Gcse Science Marked Teachers
| Topic: Determining The Enthalpy Change Of The Deposition Of Calcium Carbonate Gcse Science Marked Teachers Determine The Enthalpy Change For The Decomposition Of Calcium Carbonate |
| Content: Answer Sheet |
| File Format: PDF |
| File size: 2.6mb |
| Number of Pages: 15+ pages |
| Publication Date: July 2019 |
| Open Determining The Enthalpy Change Of The Deposition Of Calcium Carbonate Gcse Science Marked Teachers |
 |

Answered The Deposition Of Calcium Carbonate Bartle
| Topic: Answered The Deposition Of Calcium Carbonate Bartle Determine The Enthalpy Change For The Decomposition Of Calcium Carbonate |
| Content: Synopsis |
| File Format: PDF |
| File size: 1.5mb |
| Number of Pages: 35+ pages |
| Publication Date: August 2019 |
| Open Answered The Deposition Of Calcium Carbonate Bartle |
 |
Its definitely easy to prepare for determine the enthalpy change for the decomposition of calcium carbonate Calculate the enthalpy of deposition of caco3 into cao and co2the of formation of caco3 is 2885 k cal mol for cao is 1519 k cal mol and for co2is 9 an experiment to determine the enthalpy change for the deposition of calcium carbonate gcse science marked teachers 13 chapts 5 6 test determine the enthalpy change for chegg investigate the enthalpy values for the reaction between calcium carbonate and calcium oxide with hydrogen chloride gcse science marked teachers question 9 determine the enthalpy change for the chegg calculating the enthalpy change of deposition calculate the enthalpy for the formation of calcium chegg investigate the enthalpy values for the reaction between calcium carbonate and calcium oxide with hydrogen chloride gcse science marked teachers


0 Comments